
Gribskov, M., Devereux, J., and Burgess, R. (1982) Recognition of protein coding regions in DNA sequences. 
Oxford Molecular, Oxford, England.įickett, J. Oxford Molecular Group (1998) MacVector 6.5 User Guide. This process is experimental and the keywords may be updated as the learning algorithm improves. These keywords were added by machine and not by the authors. MacVector also comes with a module for contig assembly, called AssemblyLIGN. At the time of writing, the version of MacVector available was 6.5. It provides all of the most commonly used nucleic acid and protein analysis tools and also provides access via the Internet to the public Entrez databases at the National Center for Biotechnology Information (NCBI). It is an integrated comprehensive sequence analysis program that runs on the Macintosh. MacVector™, from Oxford Molecular Group, Campbell CA, is one such computer package. It is evident by now that efficiently performing the above operations on thousands or millions of base pairs by hand is so difficult as to be impossible, and computer programs that do sequence analysis are becoming more and more ubiquitous in laboratories practicing molecular biology. Getting Started with MacVector: An overview of primer design workflows in MacVector.Whether a researcher is working on a genome project, or is cloning and characterizing a gene of interest, the ability to manipulate, analyze, and annotate sequence data is becoming increasingly important.Melissa Caimano on HOW DO I video guides to common molecular biology workflows.admin on HOW DO I video guides to common molecular biology workflows.mariam abdelmalak on Major release details – Summary.Brian on Designing primers and documenting In-Fusion Cloning with MacVector.Chris on Designing primers and documenting In-Fusion Cloning with MacVector.How to call heterozygotes in trace files or Assembly Projects.MacVectorTip: How to Customize the Toolbars of MacVector windows.MacVectorTip: Selecting the sequence from a single restriction enzyme site to the end of a linear sequence.Sequence Assembly: What can Assembler do for my lab?.The window that opens contains a COPY of the starting sequence – now click on the Add Seqs button and select all the ABI/.ab1/SCF chromatogram files from your sequencing project directory to import them into the window In any event, open that sequence first, then choose Analyze | Align To Reference. It can even be the entire genome of an organism to which you want to align chromatograms from sequencing runs of mRNA or cDNA clones. Otherwise, you would normally start with a Reference sequence – it might be the predicted sequence of the PCR fragment you cloned, the starting sequence for a mutagenesis experiment or even a related sequence from another organism. I’m not going to discuss that here but there is a tutorial that you can download from this link. Admittedly, that is not always the case – if you are trying to determine the sequence of an unknown piece of DNA, then you need to use the add-on Assembler module. 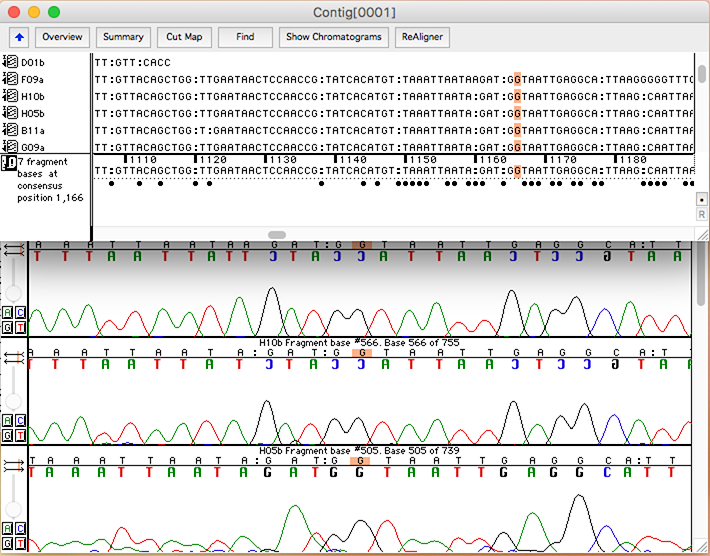
Typically, if you are aligning chromatograms it is because you are re-sequencing something. None of these algorithms will “flip” the chromatograms when required, but the strategies described below will do that. 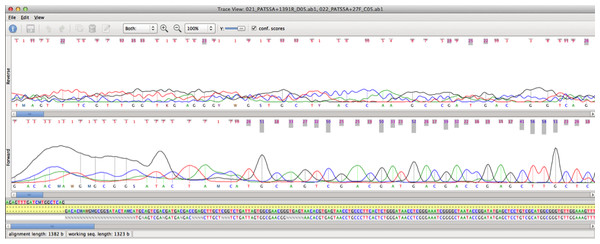
Note that you should NOT use the Multiple Sequence Alignment ( ClustalW, Muscle or T-Coffee) interface unless you are truly looking for the evolutionary relationships between the sequences and know that they are already all in the correct orientation. One common request we get is “I want to see my chromatograms/traces aligned so that I can edit the alignments”. The blog link discusses the 6 main alignment algorithms in MacVector and how to decide which is the most appropriate for accomplishing different tasks. I blogged about this a few years ago, but its something that still comes up on a regular basis.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
